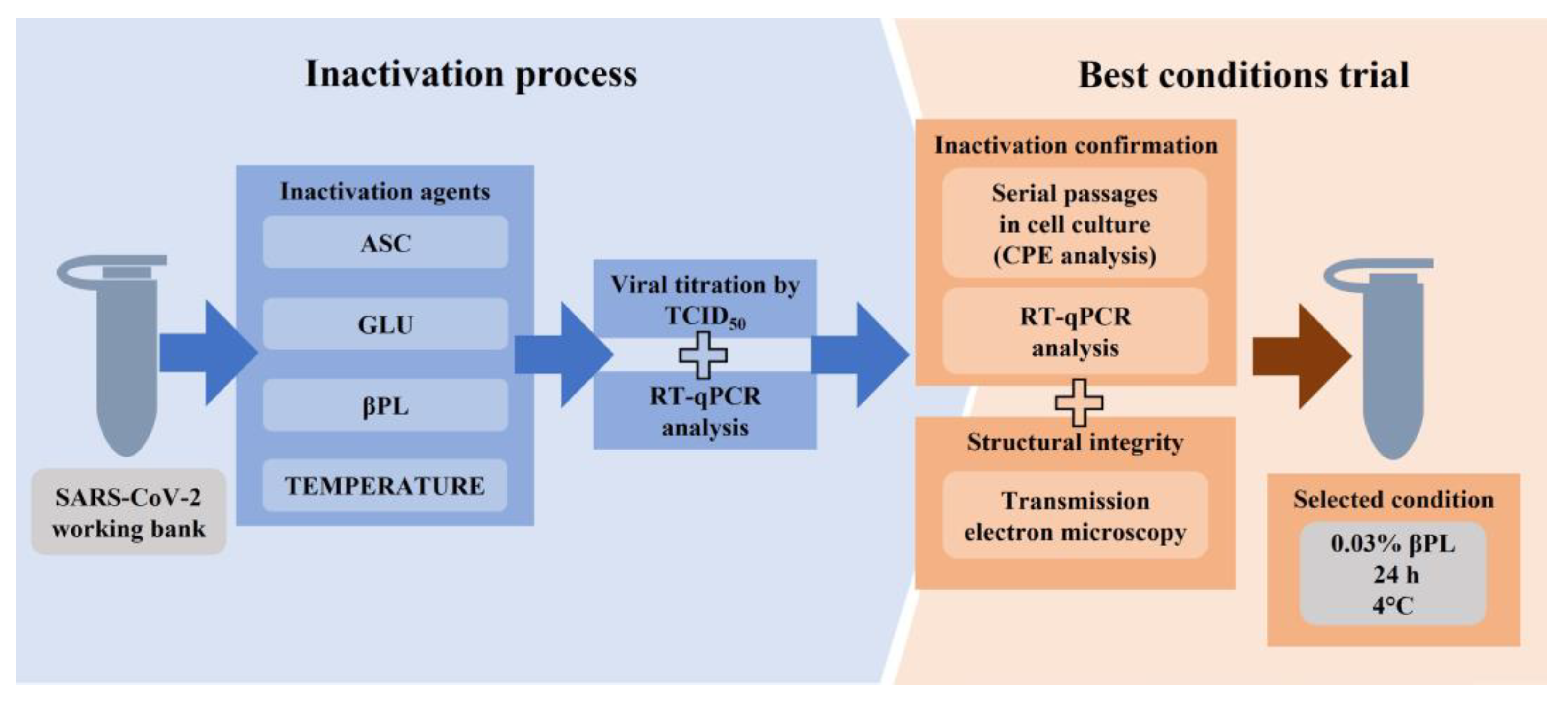
Viruses | Free Full-Text | Heat Inactivation of Different Types of SARS-CoV-2 Samples: What Protocols for Biosafety, Molecular Detection and Serological Diagnostics?
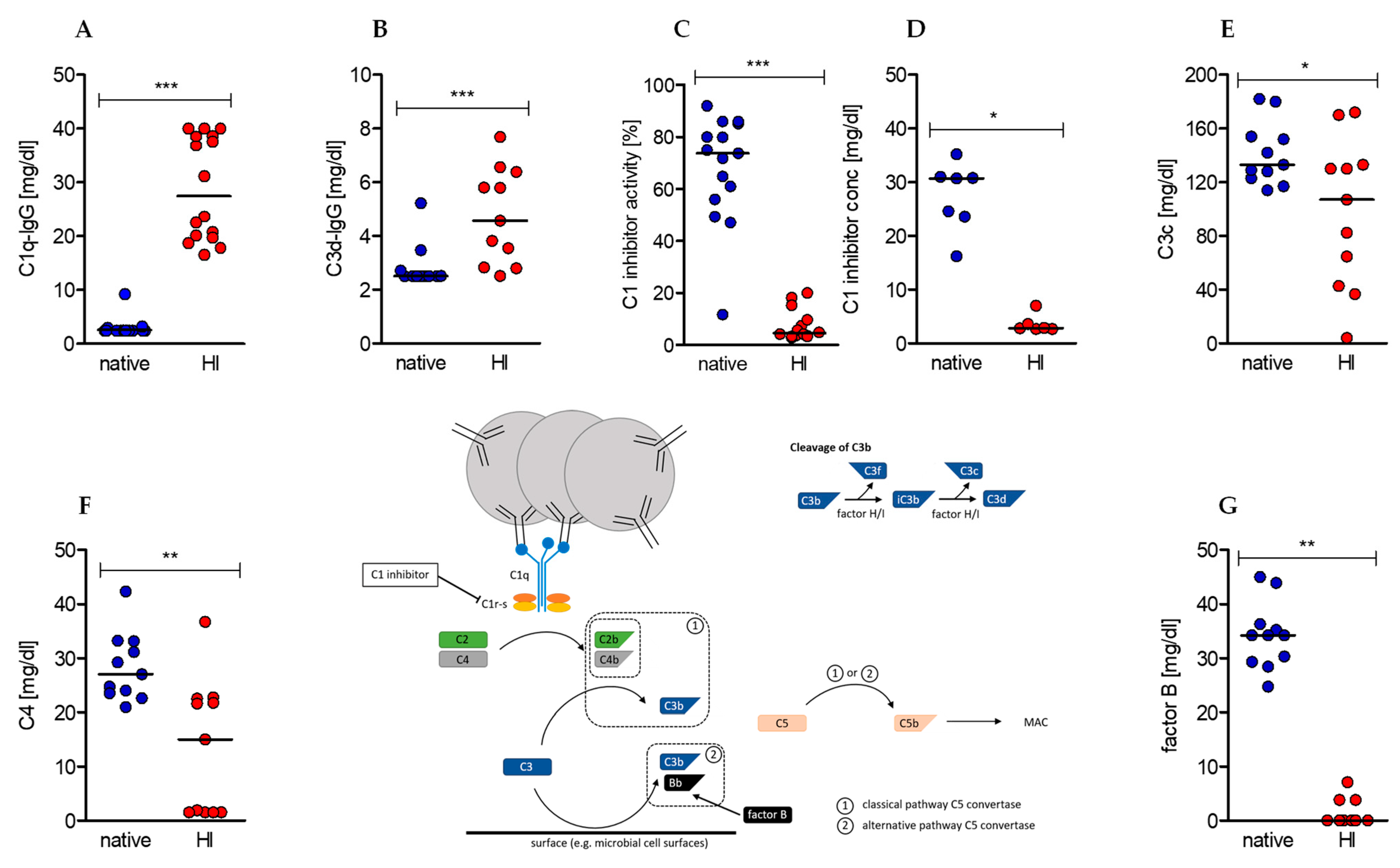
IJMS | Free Full-Text | Heat-Inactivation of Human Serum Destroys C1 Inhibitor, Pro-motes Immune Complex Formation, and Improves Human T Cell Function
Viruses | Free Full-Text | Inactivated and Immunogenic SARS-CoV-2 for Safe Use in Immunoassays and as an Immunization Control for Non-Clinical Trials
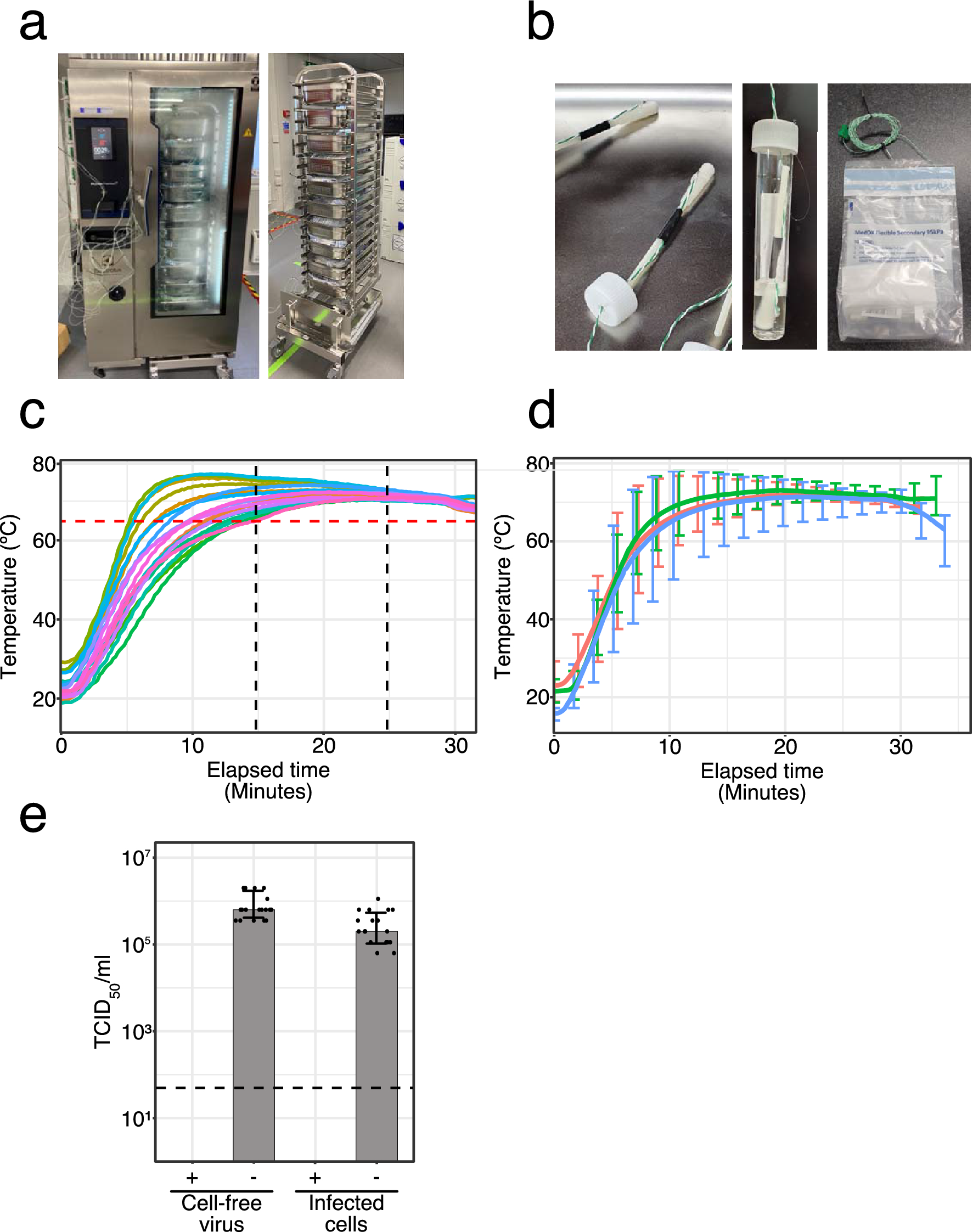
Heat inactivation of clinical COVID-19 samples on an industrial scale for low risk and efficient high-throughput qRT-PCR diagnostic testing | Scientific Reports

Round Glass Fetal Bovine Serum - Charcoal/Dextran Treated, Heat Inactivated, Capacity: 500ml at Rs 38000 in Chennai
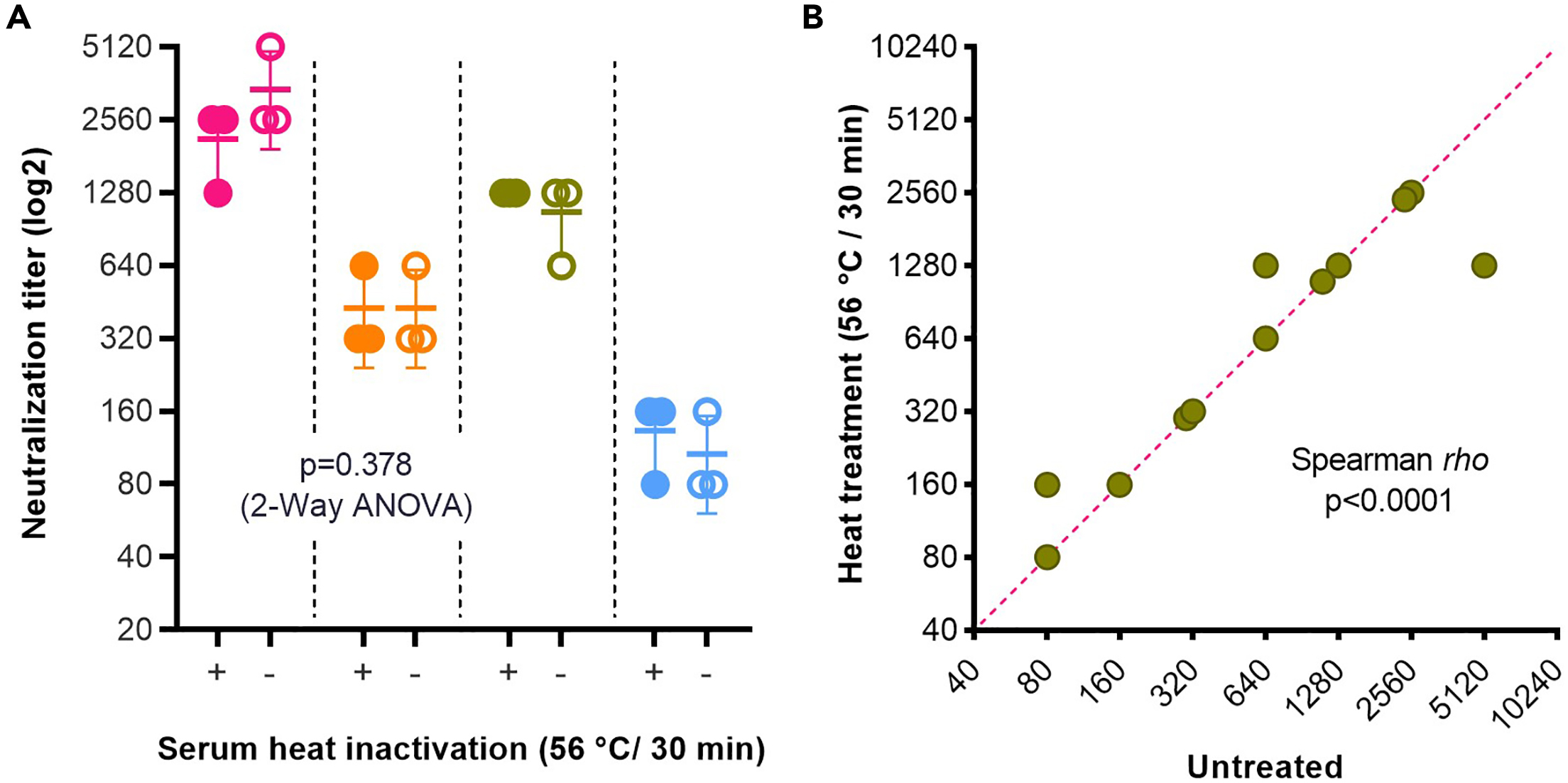
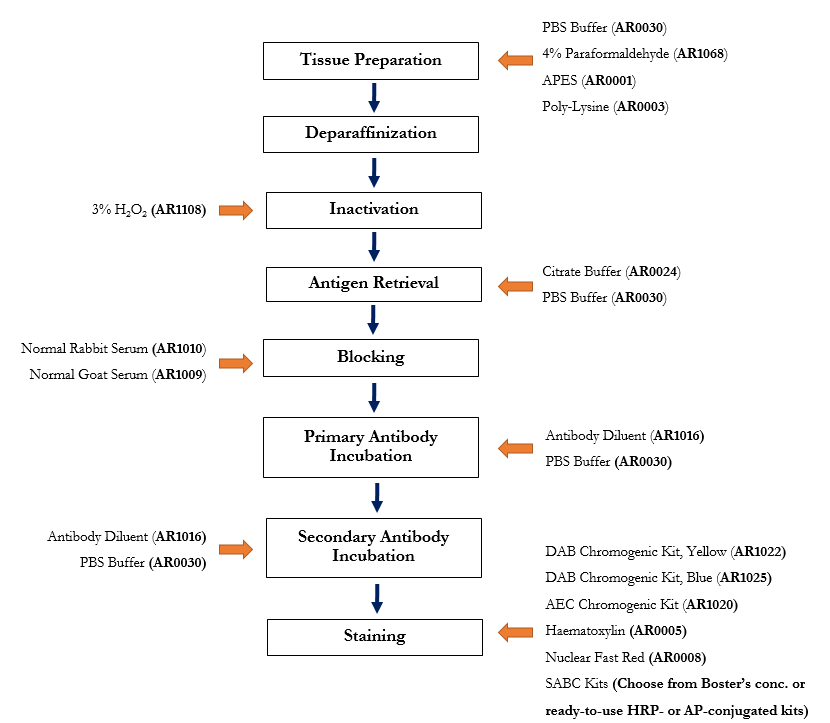
A tissue culture infectious dose-derived protocol for testing of SARS-CoV-2 neutralization of serum antibodies on adherent cells

Pathogens | Free Full-Text | Art of the Kill: Designing and Testing Viral Inactivation Procedures for Highly Pathogenic Negative Sense RNA Viruses
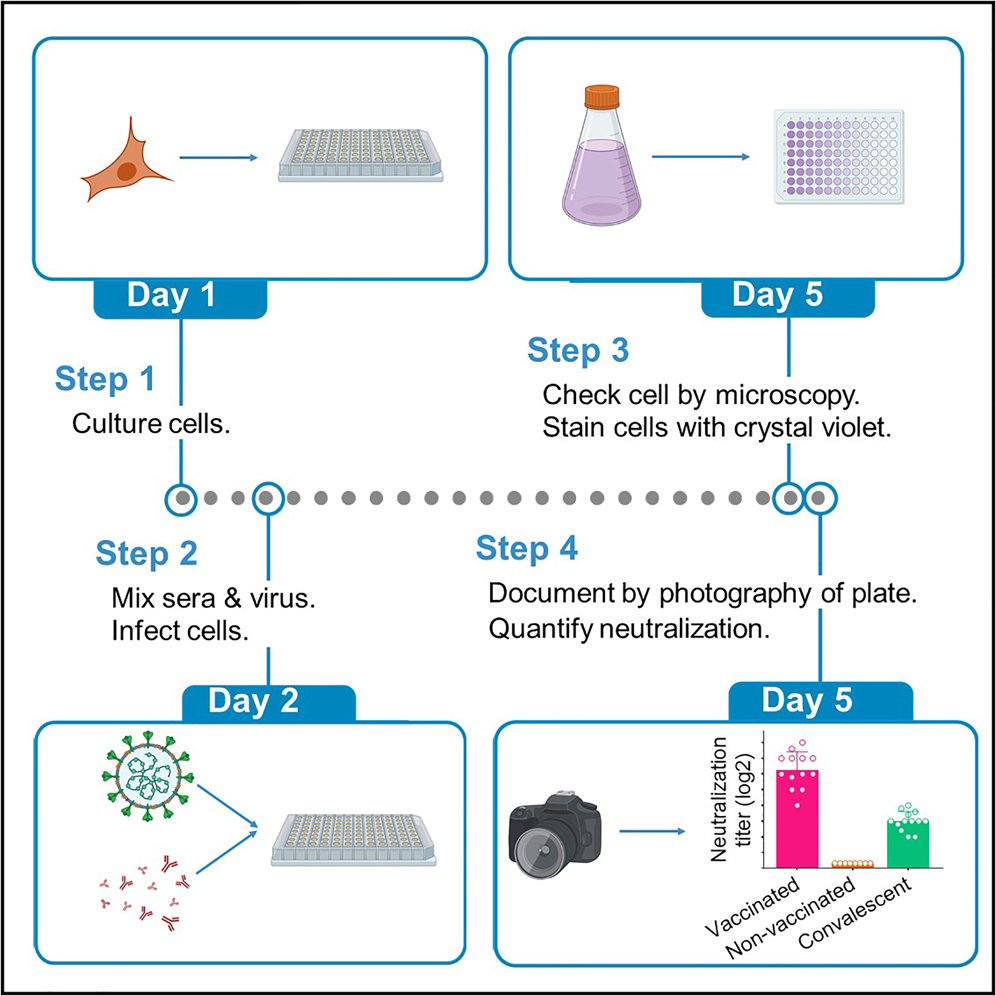
A tissue culture infectious dose-derived protocol for testing of SARS-CoV-2 neutralization of serum antibodies on adherent cells

Immunoassay-Compatible Inactivation of SARS-CoV-2 in Plasma Samples for Enhanced Handling Safety | ACS Omega

A tissue culture infectious dose-derived protocol for testing of SARS-CoV-2 neutralization of serum antibodies on adherent cells - ScienceDirect

Heat inactivation of serum interferes with the immunoanalysis of antibodies to SARS‐CoV‐2 - Hu - 2020 - Journal of Clinical Laboratory Analysis - Wiley Online Library

Endogenous complement human serum bactericidal assay (enc-hSBA) for vaccine effectiveness assessments against meningococcal serogroup B | npj Vaccines